Paxil Withdrawal
Symptoms of Withdrawal
Paxil Withdrawal Symptoms
Paxil is an antidepressant from the class of drugs under SSRI (selective serotonin reuptake inhibitor). GlaxoSmithKline (GSK), one of the most prestigious pharmaceutical companies in the world, manufactures and distributes Paxil.
Paxil has been considered as the most effective antidepressant and the most prescribed by doctors in the United States. But the moment Paxil was launched, there were many investigative studies conducted because of controversies and lawsuits surrounding the adverse effects of SSRIs. For more than ten years, GlaxoSmithKline (GSK) has marketed Paxil as non-habit forming. However, this claim was countered by a lot of court orders, expert analysis and continued investigative reports regarding Paxil and other SSRIs.
The International Federation of Pharmaceutical Manufacturers Association said that GlaxoSmithKline (GSK) has misled the consumers and suppressed the truth about Paxil regarding its numerous side effects.
Charles Medawar, the head of Social Audit, was quoted on the British Medical Journal saying something about Paxil and other SSRIs as: “This drug has been promoted for years as safe and easy to discontinue . . . The fact that it can cause intolerable withdrawal symptoms of the kind that could lead to dependence is enormously important to patients, doctors, investors and the company,” and “The company has been promoting paroxetine directly to consumers as ‘non-habit forming’ for far too long.”
GlaxoSmithKline now acknowledges the existence of possible occurrence of discontinuation syndrome, which includes severe discontinuation symptoms after taking Paxil and the other SSRIs they manufacture.
Although generally believed to be safe, discontinuation of Paxil and other SSRIs may mimic certain serious ailments and may cause intense discomfort and distress. Continuing research on the exact mechanism of Paxil and other SSRI discontinuation syndrome has attributed it to a variety of factors such as electrophysilogical changes in the body (i.e. nerve growth factor), an over-excited immune system, as well as dopamine dependency.
Paxil and other SSRI discontinuation syndrome (withdrawal syndrome) occurs when medication is interrupted, the dosage reduced or a complete cessation on using Paxil. This condition begins during the time of the dosage reduction or complete cessation, depending on the person’s metabolism and the elimination of half-life (a period of time it takes for a substance to decay or decrease by half).
With the withdrawal from Paxil, a psychoactive drug that helps the nerve cells in the brain to communicate, a certain sense of confusion occurs. Paxil side effects and discontinuation affects the other neurotransmitter systems (i.e. dopamine, norepinephrine and the g-aminobutyric acid) that are implicated in anxiety and depressive disorders. On the other hand, dependency on Paxil may result to addiction.
According to reports, it is shown that Paxil has one of the highest incidents of severe withdrawal syndrome that are due to SSRI treatment that has lasted for four or more weeks. These symptoms interfere with a person’s normal social, occupational and other aspects of life and aren’t because of a relapse of the condition before Paxil was originally prescribed by the doctor.
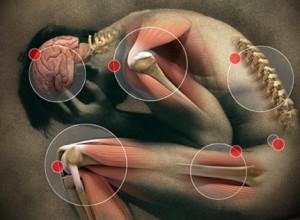
Many disputes regarding the specifics of the syndrome due to Paxil were pointed out by the users, agents, experts and doctors. Nonetheless, certain neurological symptoms occur such as headaches, somnolence or dizziness, vertigo or light-headedness, “brain shocks” (or parathesia, electric shock-like, tingling, burning, numbing and prickling sensation of a person’s skin), nausea, vomiting, sweating, insomnia, nightmares or vivid dreams, confusion/disorientation. Akathisia or restlessness can also be a symptom of withdrawal from using Paxil and other SSRIs.
Discontinued use of Paxil can cause a rebound effect with exaggerated symptoms that can take long periods of time to overcome even after a considerable amount of time had elapsed. These side effects can last for months or even years after discontinuing using Paxil. A gradual decrease of the dosage may help in the prevention of the discontinuation or withdrawal syndrome and its symptoms. Another recommended solution would be to switch from Paxil or other SSRIs to a fluoxetine (Prozac, Fontex, Seromex, Seronil, Sarafem or Fluctin), which was proven to have a longer half-life and therefore can lessen the severity of the discontinuation syndrome.
Post-SSRI sexual dysfunction (PSSD) is a type of sexual dysfunction that can be caused by Paxil and other SSRIs. Although uncommon, PSSD can last for months or even years after using Paxil. Sometimes, because of this Paxil side effect, patients are not sufficiently warned by their physicians or doctors since it is not listed in the consumer information leaflets or labels.
Discontinuation of Paxil or other SSRIs can cause the following sexual symptoms such as: Absent or decreased libido, premature ejaculation, persistent genital arousal disorder, ejaculatory anhedonia (reduced or no experience of any pleasure during orgasm), impotence or reduced vaginal lubrication, and weakened penile, vaginal, or clitoral sensitivity. Paxil and other SSRIs can also cause genital anesthesia, difficulty in maintaining an erection or getting aroused, reduced seminal volume, reduced/loss of sexual stimuli, and Priapism (persistent erectile state of the penis/clitoris).
Sudden discontinuation of Paxil or other SSRIs can also lead to adverse effects during pregnancy. It is advised by many that women who are planning to become pregnant while still under Paxil medication should avoid taking Paxil or other SSRIs to avoid exposing the fetus to harmful effects of serotonin. In some cases, pregnant women undergoing Paxil or other SSRI treatment should be individualized because discontinuing or cessation in using the drug may induce spontaneous abortion. On the other hand, Paxil can cause infants exposed to it and other SSRIs in the first trimester can have an increased risk of having cardiovascular deformities.
Discontinuation of Paxil medication during pregnancy can cause a relapse and may agitate the fetus and eventually cause a spontaneous abortion. Special cases like this recommended supervision by a doctor or physician. These recommendations are based on multiple systematic reviews and analytical reports regarding pregnant women taking Paxil or other SSRIs during pregnancy and the effects on their children or unborn fetuses.
Physical symptoms like diarrhea, constipation, tremors, fatigue, headaches, gait instability, irritability and visual impairment can also manifest during the withdrawal syndrome from Paxil and other SSRIs.
 The US FDA (Food and Drug Administration) requires Paxil to issue contraindications. Paxil carries this contraindication in its label. Because of increased risk of suicidal tendencies and other adverse effects, Paxil and other SSRIs have the black box warning, the highest type of warning in prescription drug labeling. This warning applies to all SSRIs.
The US FDA (Food and Drug Administration) requires Paxil to issue contraindications. Paxil carries this contraindication in its label. Because of increased risk of suicidal tendencies and other adverse effects, Paxil and other SSRIs have the black box warning, the highest type of warning in prescription drug labeling. This warning applies to all SSRIs.
In the United Kingdom, the Medicines and Healthcare products Regulatory Agency recommended this contraindication when several reports regarding the different Paxil side effects emerged. They urge younger individuals (people under 18 years of age) not to use Paxil and other SSRIs.
To learn more about Paxil Click Here